The Centers for Medicare and Medicaid Services (CMS) recently unveiled the 2024 Medicare Physician Fee Schedule (MPFS) final rule, bringing significant changes to the field of radiology. Here’s a concise overview of the crucial takeaways:
Conversion Factor and Overall Impact
The Centers for Medicare & Medicaid Services has released its 2024 final rule for the Medicare Physician Fee Schedule. The rule finalizes the proposed decrease to the conversion factor of -3.4%, resulting in a conversation factor of $32.74 for 2024. This is a net decrease of $1.15 to the current conversion factor of $33.89 used in 2023.
The Consolidated Appropriations Act (CCA) of 2023 provided a temporary ‘bump’ to the CF for both 2023 and 2024. The final 2024 CF reflects the expiration of the 2023 increase of +2.5% as well as the implemented +1.5% statutory increase as mandated by the CCA.
However, days before the release of the final rule, a bill moved forward in the Senate finance committee that would maintain the +2.5% CF increase from the CCA into 2024. Advocate is following the progression of this legislation closely and will provide updates as they occur.
- Final Medicare Fee Schedule CF: $32.74
- Decrease of -$1.15 from 2023 CF of $33.89
- Final Anesthesia CF: $20.43
- Decrease of -$.69 from 2023 CF of $21.12
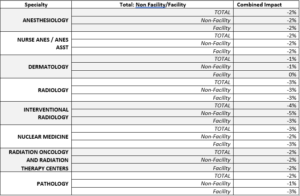
Appropriate Use Criteria/Clinical Decision Support (AUC/CDS) Program
Continual issues with implementation have led CMS to pause the PAMA Appropriate Use Criteria (AUC) program for advanced diagnostic imaging services. The real-time claims processing aspect proved to be an insurmountable obstacle. While acknowledging the value of clinical decision support, CMS encourages voluntary use of these tools. The ACR is actively working with Congress to streamline and modernize the PAMA AUC program, which includes the removal of specific requirements.
Office/Outpatient E/M Visit Complexity Add-On
HCPCS code G2211, addressing visit complexity inherent to evaluation and management associated with medical care services, is set to have an “active” status indicator starting January 1, 2024. CMS clarified the criteria for its use, emphasizing the importance of a trusting relationship between the patient and practitioner. This code is not restricted to any particular specialty.
Quality Payment Program (QPP)
In a welcome deviation from the initial proposal, CMS has decided NOT to increase the penalty threshold for the Merit-based Incentive Payment System (MIPS) for 2024. The threshold will remain at 75 points, similar to both the 2022 and 2023 performance years, rather than increasing to the proposed 82 points. This is a huge relief for providers, particularly those in specialties that have fewer ways to earn the necessary points towards their scores.
MIPS category weights for the 2024 performance year remain unchanged, with Quality at 30%, Cost at 30%, PI at 25%, and IAs at 15%. CMS will offer category reweighting for physicians unable to submit data for one or more performance categories, with the weight redistributed primarily to the Quality category.
Minimal changes were proposed to the Quality measures offered nationally via QPP, although it is important to note the final removal of three measures of particular importance for Radiology:
- 147: Nuclear Medicine
- 324: Cardiac Stress Testing on Asymptomatic Patients
These measures will no longer be available to report in 2024
Please note that measure 436: Radiation Dose Lowering was originally proposed for removal, but CMS opted to maintain the measure for one more performance year – meaning the measure will now be removed in 2025.
Additional MIPS Value Pathways (MVPs) have been approved for the upcoming year, demonstrating CMS’s continued commitment of developing an alternative reporting options for traditional MIPS.
The 2024 MPFS Final Rule brings several important changes for healthcare providers impacting pricing, regulatory programs, coding, and the Quality Payment Program. As always, ADVOCATE will keep you up to date on this and all issues impacting medical groups as they become available.
Kayley Jaquet
Manager, Regulatory Affairs